- World
- Jun 20
WHO targets antimicrobial resistance
The World Health Organisation (WHO) has launched a global campaign urging governments to adopt a tool to reduce the spread of antimicrobial resistance, adverse events and costs.
The AWaRe tool was developed by the WHO Essential Medicines List to contain rising resistance and make antibiotic use safer and more effective.
The tool, known as AWaRe, classifies antibiotics into three groups…
* Access - antibiotics used to treat the most common and serious infections
* Watch - antibiotics available at all times in the health care system
* Reserve - antibiotics to be used sparingly or preserved and used only as a last resort
The new campaign aims to increase the proportion of global consumption of antibiotics in the Access group to at least 60 per cent, and to reduce the use of antibiotics most at risk of resistance from the Watch and Reserve groups. Using Access antibiotics lowers the risk of resistance because they are ‘narrow-spectrum’ antibiotics (that target a specific microorganism rather than several). They are also less expensive because they are available in generic formulations.
Background
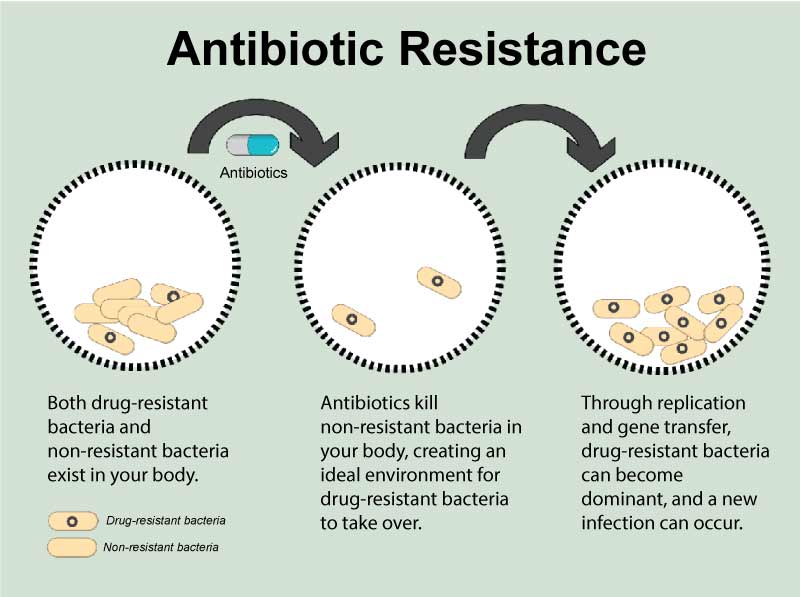
* Antimicrobial resistance is a global health and development threat that continues to escalate globally, as highlighted in a recent report by the International Coordination Group on Antimicrobial Resistance.
* Currently, it is estimated that more than 50 per cent of antibiotics in many countries are used inappropriately, such as for treatment of viruses when they only treat bacterial infections or use of the wrong (broader spectrum) antibiotic, thus contributing to the spread of antimicrobial resistance.
* One of the most pressing concerns is the spread of resistant gram-negative bacteria, including Acinetobacter, Escherichia coli and Klebsiella pneumoniae. These bacteria, which are commonly seen in hospitalised patients, cause infections like pneumonia, bloodstream infections, wound or surgical site infections and meningitis. When antibiotics stop working effectively, more expensive treatments and hospital admissions are needed, taking a heavy toll on already stretched health budgets.
* At the same time, many low- and middle-income countries experience vast gaps in access to effective and appropriate antibiotics.
* Childhood deaths due to pneumonia (estimated globally at close to 1 million per year) because of lack of access to antibiotics remain frequent in many parts of the world. And although more than 100 countries have put in place national plans to tackle antimicrobial resistance, only about one-fifth of those plans are funded and implemented.
* According to a UN report, drug-resistant diseases could cause 10 million deaths each year by 2050 and damage to the economy as catastrophic as the 2008-09 global financial crisis. By 2030, antimicrobial resistance could force up to 24 million people into extreme poverty.
Reasons for spread of antimicrobial resistance
Antibiotic consumption
The main origin of resistance to antibiotics is their misuse. As underlined by the European Centre for Disease Prevention and Control (ECDC), there are three main types of misuse…
* The unnecessary prescription of antibiotics for viral infections like flu, influenza, against which they have no effect;
* The too frequent prescription of “broad-spectrum antibiotics” in place of a better targeted antibiotic, through more precise diagnosis;
* The inadequate use by the patient, not respecting either dosage or duration of the treatment, which means that some of the bacteria may survive and become resistant.
* Indiscriminate use of antibiotic FDCs even without the knowledge of a proven advantage over single compounds, leading to emergence of bacterial strains resistant to multiple antibiotics. Approximately 118 antibiotic FDCs are available in India.
Social factors
Among the general public, social factors include self-medication, access to antibiotics without prescription, use of pharmacies and informal health care providers as sources of health care, and lack of knowledge about when to use antibiotics. the practice of over-the-counter dispensation of antibiotics without any prescription is common.
Animal protein
Although direct antibiotic sales data in food animals are not available for India, it is estimated that India was the fifth largest consumer of antibiotics in food animals (poultry, pigs and cattle) in 2010. Antibiotics such as colistin, tetracycline, doxycycline and ciprofloxacin, which are critical to human health, are commonly used for growth promotion in poultry.
Cultural factors
One of the major cultural activities associated with potential acquisition and spread of antibiotic-resistant bacteria is mass bathing in rivers as part of religious mass gathering occasions. For example, according to a study, mass bathing in the Ganga during pilgrimages may be contributing to antimicrobial resistance.
Pharma industry pollution
Wastewater effluents from the antibiotic manufacturing units contain a substantial amount of antibiotics, leading to contamination of rivers and lakes. In India, the Central Pollution Control Board established effluent standards for pharma industry waste, and all state pollution control boards use the same standards. The current standards do not include antibiotic residues, and thus they are not monitored in the pharma industry effluents.
Sanitation
Poor sanitation plays a major role in the spread of antibiotic-resistant bacteria and ARGs. According to the World Bank, more than 50 per cent of the Indian population does not have access to sanitation facilities for safe disposal of human waste. In addition, a large proportion of sewage is disposed untreated into receiving water bodies, leading to gross contamination of rivers with antibiotic residues, antibiotic-resistant organisms.
Poor infection control practices
The prevalence of various health care associated infections among Indian hospitals ranges from 11 to 83 per cent, in contrast to the WHO estimate of about 7 to 12 per cent.
This is primarily poor infection control practices. For example, a study in Mangalore, which assessed hand-washing practices of nurses and doctors, found that only 31.8 per cent of them washed hands after contact with patients.
Chennai declaration
In a bid to create a roadmap to tackle antimicrobial resistance, Indian health care experts met in 2012. All stakeholders, including representatives of medical societies, various government bodies, media, academics and international representatives, came under one roof to discuss the issue. The aim was to formulate implementable recommendations to tackle antimicrobial resistance. The meeting led to the creation of a document titled The Chennai declaration.
Salient features
* Formulation of an effective national policy to control the rising trend of antimicrobial resistance.
* Changes in the medical education curriculum to include training on antibiotic usage and infection control.
* Setting up of a national task force to guide and supervise the regional and state infection control committees.
* An Infection Control Team (ICT) be made mandatory at all hospitals.
* Regulatory authorities and accreditation agencies such as the National Accreditation Board for Hospitals and ISO must insist on a functioning ICT during the licensing and accreditation process.
* National Accreditation Board for Hospitals & Healthcare Providers (NABH) to insist on strict implementation of hospital antibiotic and infection control policy, during hospital accreditation and re-accreditation processes.
Actions taken
* To meet the obligations of the declaration, the National Programme on Containment of Antimicrobial Resistance was launched under the 12th Five-year Plan.
• The Drugs and Cosmetic Rules, 1945, were amended in 2013 to incorporate a new Schedule H1. It contains certain third and fourth generation antibiotics, certain habit forming drugs and anti-TB drugs.
• This rule is meant to regulate over-the-counter dispensing of drugs. Pharmacists not only have to insist on a prescription from a registered medical practitioner, but they also need to enter details in a register. Drug inspectors will monitor compliance.
• First-line antibiotics will not come under strict monitoring as those are excluded from the list, at least initially. The new H1 list is based on a step-by-step strategy of the Chennai declaration.
• Under the Drugs & Cosmetics Rules, drugs specified under Schedules H and X are required to be sold by retail on the prescription of a registered medical practitioner only.